Gather the Key Documents
1. Identify the full drug name(s).
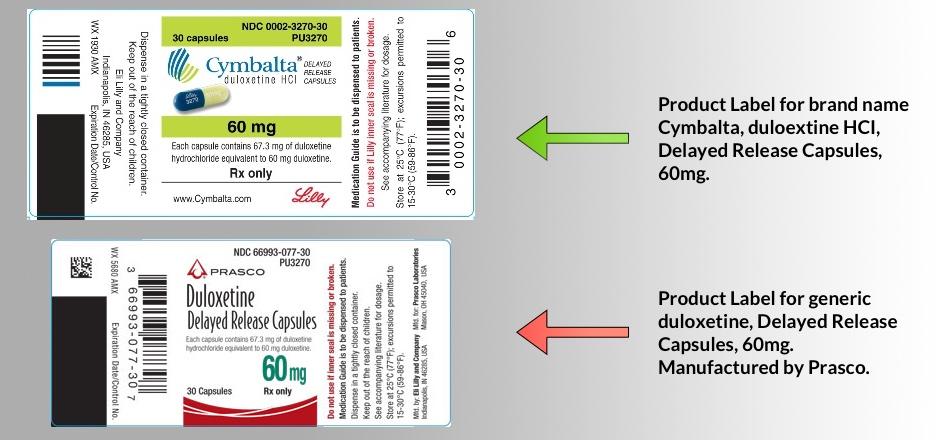
Get your drug bottle, drug packaging, and/or a copy of the drug product label that came with your drug for all of the psychiatric drugs that you are currently taking.
Make note of each drug’s official name, including whether it displays both a brand name and a generic name or just the generic name, and whether there are any supplemental aspects to the name such as acronyms or formulation descriptions (e.g. “Adderall XR” or “Depakote – Divalproex Sodium Delayed-release Tablets”). Ensure that you have recorded this name fully and accurately – it is the ‘key’ to learning more about your drug(s). If you have any doubt at all, ask your pharmacist. You will use this full drug name to obtain the other documents described in this step. If yours is a generic drug, it may also be helpful to identify the packager and/or manufacturer listed on the packaging product label.
2. Get a copy of TWP’s Guide to the FDA-approved Drug Label.
Open up "TWP’s Guide to the FDA-approved Drug Label” in a new browser tab or window. You may want to keep a copy handy by saving it as a file to your computer, selecting and copying the text on the page to a file, and/or printing it. This guide explains what information is typically in FDA-approved drug labels and highlights the topics most relevant for tapering.
3. Get a copy of the drug label(s).
The most important documents are the complete FDA-approved drug labels for all of the psychiatric drugs that you are taking, regardless of which ones you’re planning to taper off. To do this, follow the instructions and links provided in "TWP’s Guide to the FDA-approved Drug Label".
4. Get a copy of TWP’s Key to Withdrawal-related Information in the Drug Label.
Open up “TWP’s Key to Withdrawal-related Information in the Drug Label” in a new browser tab or window. This “Key” highlights some of the information in the FDA-approved drug label that can be particularly important for withdrawal. It explains the relevance of the information under a variety of “Topics”, and also shows where in the drug label this information can often be found. Again, for ease of use moving ahead, you may want to save or print a copy of this page.
5. Create a “Drug Self-education Sheet”.
Get a blank sheet of paper (or a few sheets, depending on how large your hand-writing is) and pen. On the sheet(s) of paper, write down the titles of the 'Topics' from "TWP's Key to Withdrawal-related Information in the Drug Label", leaving plenty of space in between each topic heading so that you can write down what you learn about your drug. This will be your “Drug Self-education Sheet”.
We strongly recommend that you read through all of these documents that you have gathered, and fill out what you learn about your drug on your “Drug Self-education Sheet”. However, it’s not absolutely necessary to do it all in one sitting; what’s important is to have these documents on hand and to start to become more familiar with the information in your drug labels if you have not read them before. Then, as particular issues become crucial for planning the safest possible taper in later steps of this Companion Guide, we will be directing you back to your drug label(s) and your "Drug Self-education Sheet".
In this section
- Step 10- Get Informed About Your Psychiatric Drug
- Step 11- Ensuring that a Drug is Relatively ‘Taper-friendly’
- Step 12- Interactions, Reactions and Sensitivities
- Step 13- Taper Rates
- Step 14- Taper Schedules
- Step 15- Taper Methods
- Step 16- Preparatory Decisions
- Step 17- Gather the Gear
- Step 18- Essential Skills
- Step 19- Setting Up a Taper Journal
- Step 20- Implementing a Taper
TWP’s Companion Guide to Psychiatric Drug Withdrawal Part 2: Taper